Isomaltase
Isomaltase[edit]
Isomaltase, also known as alpha-glucosidase, is an important enzyme in the digestive system that plays a crucial role in the breakdown of carbohydrates. It is part of the sucrase-isomaltase complex, which is located on the brush border of the small intestine.
Structure[edit]
Isomaltase is a component of the sucrase-isomaltase complex, which is a type of glycoprotein. This complex is composed of two subunits: sucrase and isomaltase. Each subunit has its own active site and specific substrate affinity. The isomaltase subunit is responsible for hydrolyzing alpha-1,6-glycosidic bonds found in isomaltose and other oligosaccharides.
Function[edit]
The primary function of isomaltase is to catalyze the hydrolysis of isomaltose into two molecules of glucose. This reaction is essential for the proper digestion and absorption of carbohydrates in the diet. Isomaltase also acts on other substrates, including maltose and maltotriose, contributing to the overall breakdown of dietary starches.
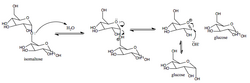
Mechanism[edit]
The enzymatic mechanism of isomaltase involves the cleavage of the alpha-1,6-glycosidic bond. This process is facilitated by the active site of the enzyme, which binds to the substrate and stabilizes the transition state, allowing for the efficient conversion of isomaltose into glucose. The diagram on the right illustrates the mechanism of action of the sucrase-isomaltase complex.
Clinical Significance[edit]
Deficiency in isomaltase activity can lead to congenital sucrase-isomaltase deficiency (CSID), a condition characterized by the inability to properly digest certain sugars, leading to symptoms such as diarrhea, abdominal pain, and bloating. Diagnosis of CSID is typically confirmed through enzyme activity assays or genetic testing.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian