Isopimaric acid
Isopimaric Acid[edit]
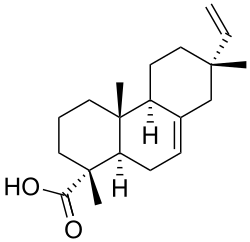
Isopimaric acid is a naturally occurring diterpene resin acid found in the oleoresin of various conifer species. It is a member of the pimarane family of diterpenes and is known for its role in the defense mechanisms of trees against herbivores and pathogens.
Chemical Structure[edit]
Isopimaric acid has the molecular formula C20H30O2. It is characterized by a bicyclic structure with a carboxylic acid functional group. The structure of isopimaric acid is similar to other resin acids such as abietic acid and pimaric acid, but it differs in the configuration of its carbon skeleton.
Biosynthesis[edit]
Isopimaric acid is synthesized in conifers through the mevalonate pathway, which is a key metabolic pathway that produces isoprenoids. The biosynthesis involves the cyclization of geranylgeranyl pyrophosphate (GGPP) to form the pimarane skeleton, followed by oxidation and rearrangement to yield isopimaric acid.
Biological Role[edit]
In conifers, isopimaric acid is a component of the oleoresin, a sticky substance that exudes from the bark and wood when the tree is injured. This resin acts as a physical barrier and contains compounds that are toxic or deterrent to insects and pathogens. Isopimaric acid contributes to the antimicrobial and insect-repellent properties of the resin.
Applications[edit]
Isopimaric acid and its derivatives have been studied for their potential applications in various fields. They have shown promise as antimicrobial agents, anti-inflammatory agents, and in the synthesis of biodegradable polymers.
Related Compounds[edit]
Isopimaric acid is closely related to other resin acids such as abietic acid, pimaric acid, and sandaracopimaric acid. These compounds share similar biosynthetic pathways and structural features, but differ in their specific chemical configurations and biological activities.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian