Lysergol
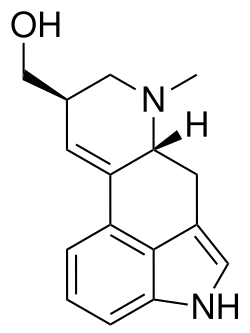
Lysergol is a naturally occurring ergoline alkaloid found in various species of the Convolvulaceae and Clavicipitaceae families. It is structurally related to other ergoline derivatives such as ergotamine and lysergic acid diethylamide (LSD).
Chemical Structure and Properties[edit]
Lysergol has the chemical formula C16H17N3O and a molecular weight of 267.33 g/mol. It features a tetracyclic ergoline ring system, which is characteristic of compounds in this class. The structure includes a fused indole ring, which is common among many biologically active compounds.
Sources[edit]
Lysergol is primarily found in the seeds of certain species of the Convolvulaceae family, such as Ipomoea and Rivea corymbosa. It is also present in the Clavicipitaceae family, particularly in the ergot fungus, which infects cereal grains like rye.
Pharmacology[edit]
Lysergol exhibits a range of pharmacological activities due to its interaction with various neurotransmitter receptors. It has been studied for its potential effects on the serotonin (5-HT) receptors, similar to other ergoline derivatives. However, its exact mechanism of action and potential therapeutic uses are not as well-documented as those of other ergolines like ergotamine and LSD.
Potential Uses[edit]
While lysergol itself is not widely used in medicine, its structural similarity to other ergoline compounds suggests potential applications in the treatment of conditions such as migraine and Parkinson's disease. Further research is needed to fully understand its pharmacological profile and therapeutic potential.
Safety and Toxicity[edit]
The safety profile of lysergol is not well-established. As with other ergoline alkaloids, there may be risks of toxicity, particularly with high doses or prolonged use. Symptoms of ergoline toxicity can include nausea, vomiting, vasoconstriction, and hallucinations.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian