Hexamethylenetetramine
Hexamethylenetetramine (also known as methenamine, urotropin, or hexamine) is a heterocyclic organic compound with the formula (CH₂)₆N₄. It is a white crystalline compound highly soluble in water and polar organic solvents.
History[edit]
Hexamethylenetetramine was first discovered by Aleksandr Butlerov in 1859. It has since been used in various applications, including as a medication, a food additive, and in the production of phenolic resins.
Chemical Structure and Properties[edit]
Hexamethylenetetramine is composed of a six-membered ring containing four nitrogen atoms and six methylene groups. Its molecular structure can be described as a cage-like structure similar to adamantane. The compound is stable under normal conditions but can decompose at high temperatures to release formaldehyde and ammonia.
Synthesis[edit]
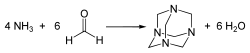
Hexamethylenetetramine is synthesized by the reaction of formaldehyde and ammonia:
Failed to parse (syntax error): {\displaystyle 4 CH₂O + 6 NH₃ → (CH₂)₆N₄ + 6 H₂O}
This reaction is typically carried out in an aqueous solution.
Applications[edit]
Medical Use[edit]
Hexamethylenetetramine is used as an antibiotic under the name methenamine. It is particularly effective in the treatment of urinary tract infections (UTIs). The compound works by decomposing into formaldehyde in acidic environments, which has antibacterial properties.
Industrial Use[edit]
Hexamethylenetetramine is used in the production of phenolic resins, which are important in the manufacture of molding compounds, laminates, and adhesives. It is also used as a fuel in solid fuel tablets for camping stoves.
Food Additive[edit]
In the food industry, hexamethylenetetramine is used as a preservative and is listed under the E number E239.
Safety and Handling[edit]
Hexamethylenetetramine is generally considered safe when used in accordance with prescribed guidelines. However, it can be irritating to the skin, eyes, and respiratory system. Proper safety measures, including the use of personal protective equipment, should be followed when handling the compound.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian