Nitro compound
Nitro compounds are organic compounds that contain one or more nitro functional groups (-NO2). These groups are attached to a carbon atom in a molecule. Nitro compounds are a diverse class of organic compounds with a wide range of applications and properties, from explosives to pharmaceuticals. The nitro group is electron-withdrawing, which significantly affects the chemical behavior of the compound.
Chemistry[edit]
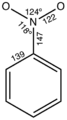
Nitro compounds are characterized by the presence of the nitro group, which consists of one nitrogen atom bonded to two oxygen atoms (N=O>O). This group is typically bonded to a carbon atom in an organic compound. The presence of the nitro group can significantly alter the chemical and physical properties of the molecule, making nitro compounds highly reactive in some cases.
Synthesis[edit]
Nitro compounds can be synthesized through various chemical reactions. One common method is the nitration of aromatic compounds using nitric acid, often in the presence of sulfuric acid as a catalyst. This process involves the substitution of a hydrogen atom on the aromatic ring with a nitro group. Aliphatic nitro compounds can also be synthesized through the reaction of alkenes with nitric acid or through the nitration of alkanes.
Reactions[edit]
Nitro compounds undergo a range of chemical reactions. They can be reduced to amines, which is a critical step in the synthesis of many pharmaceuticals and dyes. Nitro compounds can also participate in nucleophilic substitution reactions, where the nitro group is replaced by other groups. Additionally, the presence of the nitro group can facilitate the formation of other functional groups, such as carbonyls, through specific oxidation reactions.
Applications[edit]
Nitro compounds have a wide range of applications in various fields. In the pharmaceutical industry, nitro compounds are precursors to a variety of drugs, including antibiotics and analgesics. In the agricultural sector, some nitro compounds serve as herbicides and insecticides. Nitro compounds are also used in the production of dyes, polymers, and explosives. Nitroglycerin, a well-known nitro compound, is used both as a heart medication and as an explosive.
Safety and Environmental Impact[edit]
Nitro compounds, especially those used as explosives, are handled with caution due to their potential for detonation. Proper storage and handling procedures are essential to prevent accidental explosions. In the environment, nitro compounds can be toxic to aquatic life and may contribute to water and soil pollution. Bioremediation and chemical degradation are methods used to mitigate the environmental impact of nitro compounds.
See Also[edit]
-
Nitro group structure
-
Phenyl nitro compound with metric dimensions
-
Benzene structure
-
Nitronium ion
-
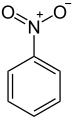
Nitrobenzene structure
-
Ter Meer reaction
-
Nitrohydrogenation process
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian