Normustine
An overview of the chemotherapy drug Normustine
Normustine is a chemical compound that belongs to the class of nitrosoureas, which are used in chemotherapy for the treatment of various types of cancer. It is known for its ability to alkylate DNA, thereby interfering with the replication of cancer cells.
Chemical Structure and Properties[edit]
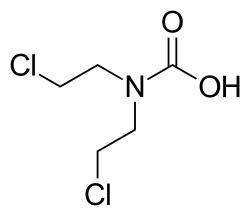
Normustine is characterized by its nitrosourea structure, which includes a nitroso group attached to a urea moiety. This structure is crucial for its function as an alkylating agent. The chemical formula of Normustine is C9H16N3O3Cl, and it has a molecular weight of 249.7 g/mol.
Mechanism of Action[edit]
Normustine exerts its antineoplastic effects primarily through the alkylation of DNA. This process involves the transfer of alkyl groups to the DNA molecule, leading to cross-linking of DNA strands. Such cross-linking prevents the normal separation of DNA strands during cell division, thereby inhibiting the replication of cancer cells. This mechanism is similar to other alkylating agents used in chemotherapy.
Clinical Use[edit]
Normustine is used in the treatment of various malignancies, including brain tumors, lymphomas, and melanoma. Its ability to cross the blood-brain barrier makes it particularly useful in treating brain cancers. The drug is typically administered in combination with other chemotherapeutic agents to enhance its efficacy.
Side Effects[edit]
As with many chemotherapy drugs, Normustine can cause a range of side effects. Common adverse effects include myelosuppression, which is a decrease in bone marrow activity leading to reduced production of blood cells. This can result in anemia, increased risk of infection, and bleeding. Other side effects may include nausea, vomiting, and fatigue.
Pharmacokinetics[edit]
Normustine is administered intravenously and is rapidly distributed throughout the body. It is metabolized in the liver and excreted primarily through the kidneys. The drug's ability to penetrate the blood-brain barrier is a significant advantage in treating central nervous system malignancies.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian