Omadacycline
What is Omadacycline?[edit]
- It is an aminomethylcycline which is a semisynthetic derivative of the tetracycline class of antibacterial drugs, for intravenous or oral administration.
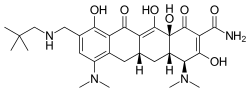
- Chemical name: (4S,4aS,5aR,12aS)-4,7-bis(dimethylamino)-9-(2,2-dimethylpropylaminomethyl)-3,10,12,12a-tetrahydroxy-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide, 4-methylbenzenesulfonate.
- Molecular formula: C36H48N4O10S (monotosylate salt).
- Molecular weight: 728.9 (monotosylate salt).
What are the uses of this medicine?[edit]
- It is indicated for the treatment of adult patients with community-acquired bacterial pneumonia (CABP) caused by the following susceptible microorganisms: Streptococcus pneumoniae, Staphylococcus aureus (methicillin-susceptible isolates), Haemophilus influenzae, Haemophilus parainfluenzae, Klebsiella pneumoniae, Legionella pneumophila, Mycoplasma pneumoniae, and Chlamydophila pneumoniae.
- It is indicated for the treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by the following susceptible microorganisms: Staphylococcus aureus (methicillin-susceptible and -resistant isolates), Staphylococcus lugdunensis, Streptococcus pyogenes, Streptococcus anginosus grp. (includes S. anginosus, S. intermedius, and S. constellatus), Enterococcus faecalis, Enterobacter cloacae, and Klebsiella pneumoniae.
How does this medicine work?[edit]
Omadacycline is an aminomethylcycline antibacterial within the tetracycline class of antibacterial drugs. Omadacycline binds to the 30S ribosomal subunit and blocks protein synthesis. In general, omadacycline is considered bacteriostatic; however, omadacycline has demonstrated bactericidal activity against some isolates of S. pneumoniae and H. influenzae.
Who Should Not Use this medicine?[edit]
It is contraindicated in patients with known hypersensitivity to omadacycline or tetracycline-class antibacterial drugs, or to any of the excipients.
What are the brand names and dosage forms of this medicine?[edit]
- Brand name: Nuzyra; NDC Codes: 71715-001-01, 71715-001-02, 71715-002-21, 71715-002-23, 71715-002-24, 71715-002-27, 71715-002-28.
- NUZYRA for Injection: Each single-dose vial contains 100 mg omadacycline (equivalent to 131 mg omadacycline tosylate) which must be reconstituted and further diluted prior to intravenous infusion. The lyophilized powder is a yellow to dark orange cake.
- NUZYRA Tablets: Each tablet contains 150 mg of omadacycline (equivalent to 196 mg omadacycline tosylate) in yellow, diamond-shaped, film-coated tablets debossed with OMC on one side and 150 on the other side.
Is this medicine FDA approved?[edit]
It is approved by US FDA on October 2, 2018.
What are the active and inactive ingredients in Omadacycline?[edit]
- For injection:
- Active ingredients: omadacycline
- Inactive ingredients: Sucrose, Hydrochloric Acid, Sodium Hydroxide, Water.
- For tablet:
- Active ingredients: omadacycline
- Inactive ingredients: silicon dioxide, crospovidone, glyceryl monocaprylocaprate, ferric oxide yellow, lactose monohydrate, microcrystalline cellulose, polyvinyl alcohol, unspecified sodium bisulfite, sodium lauryl sulfate, sodium stearyl fumarate, talc, titanium dioxide.
How should this medicine be used?[edit]
- NUZYRA for Injection: Do NOT administer NUZYRA for injection with any solution containing multivalent cations, e.g., calcium and magnesium, through the same intravenous line. Co-infusion with other medications has not been studied.
- NUZYRA Tablets: Fast for at least 4 hours and then take with water. After oral dosing, no food or drink (except water) is to be consumed for 2 hours and no dairy products, antacids, or multivitamins for 4 hours.
What side effects can this medication cause?[edit]
- Mortality Imbalance in Patients with Community-Acquired Bacterial Pneumonia
- Tooth Development and Enamel Hypoplasia
- Inhibition of Bone Growth
- Hypersensitivity Reactions
- Tetracycline Class Effects
What to do in case of emergency/overdose?[edit]
- No specific information is available on the treatment of overdosage with NUZYRA. Following a 100 mg single dose intravenous administration of omadacycline, 8.9% of dose is recovered in the dialysate.
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
NUZYRA, like other tetracycline-class antibacterial drugs, may cause discoloration of deciduous teeth and reversible inhibition of bone growth when administered during the second and third trimester of pregnancy. So, it cannot be used in pregnancy.
Can this medicine be used in children?[edit]
- Safety and effectiveness of NUZYRA in pediatric patients below the age of 18 years have not been established.
- Due to the adverse effects of the tetracycline-class of drugs, including NUZYRA on tooth development and bone growth, use of NUZYRA in pediatric patients less than 8 years of age is not recommended
What should I know about storage and disposal of this medication?[edit]
NUZYRA for Injection and NUZYRA Tablets should be stored at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). Do not Freeze.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
