Pafuramidine
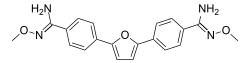
Pafuramidine (also known as DB289 and DB75) is a synthetic, orally-active prodrug of Furamidine. It was developed by Immtech Pharmaceuticals and was in Phase III clinical trials for the treatment of Human African trypanosomiasis, also known as sleeping sickness, and Pneumocystis pneumonia (PCP) in patients with HIV/AIDS. However, the development was halted due to safety concerns.
Chemistry[edit]
Pafuramidine is a diamidine compound, specifically a prodrug of Furamidine. It is designed to improve the bioavailability and reduce the toxicity of the parent drug. The conversion of Pafuramidine to Furamidine occurs in the liver.
Pharmacology[edit]
Pafuramidine exhibits its activity by being metabolized to its active form, Furamidine. Furamidine then interferes with the DNA of the parasite, leading to its death. It is active against both the early (hemolymphatic) and late (neurological) stages of Human African trypanosomiasis.
Clinical Trials[edit]
Pafuramidine was in Phase III clinical trials for the treatment of Human African trypanosomiasis and Pneumocystis pneumonia in patients with HIV/AIDS. However, the trials were halted due to safety concerns, specifically potential kidney toxicity.
Safety[edit]
During the clinical trials, some patients developed kidney toxicity, which led to the halting of the trials. The exact cause of this toxicity is not known, but it is believed to be related to the metabolism of Pafuramidine in the body.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
