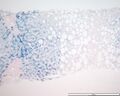
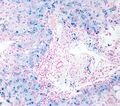
Perls Prussian blue
Perls' Prussian Blue is a histochemical staining method used in Histology and Pathology to detect the presence of Iron deposits in tissues. This technique is particularly useful in diagnosing conditions associated with abnormal iron accumulation, such as Hemochromatosis, Hemosiderosis, and certain types of Anemia. The stain, which is also known as Prussian blue or Iron blue, binds to ferric iron (Fe3+) in tissues, resulting in a distinctive blue coloration that can be observed under a microscope.
History[edit]
The method was developed in the 19th century by the German pathologist Max Perls, who discovered that potassium ferrocyanide reacts with ferric iron in acidic solutions to form a blue pigment, ferric ferrocyanide. This reaction forms the basis of the Perls' Prussian Blue staining procedure.
Procedure[edit]
The Perls' Prussian Blue staining procedure involves treating tissue sections with a mixture of hydrochloric acid and potassium ferrocyanide. This mixture reacts with ferric iron present in the tissue to form ferric ferrocyanide, a blue pigment. The stained sections are then observed under a light microscope. Iron deposits appear as blue granules within the tissue, allowing for the identification and localization of iron accumulation.
Applications[edit]
Perls' Prussian Blue staining is widely used in medical diagnostics to identify and assess the severity of diseases associated with iron overload or abnormal iron metabolism. It is particularly important in the diagnosis of:
- Hemochromatosis: A genetic disorder causing excessive iron absorption and accumulation in the body.
- Hemosiderosis: A condition characterized by the accumulation of hemosiderin, an iron-storage complex, due to bleeding or iron overload.
- Anemia: Certain types of anemia, such as sideroblastic anemia, can be associated with abnormal iron deposits in the bone marrow.
Limitations[edit]
While Perls' Prussian Blue staining is a valuable tool for detecting iron in tissues, it has some limitations. It is specific for ferric iron (Fe3+) and does not detect ferrous iron (Fe2+). Additionally, the technique requires careful handling of hazardous chemicals and precise control of staining conditions to achieve accurate results.
See Also[edit]
-
Siderophage iron stain in CSF
-
Hemochromatosis liver iron stained with Prussian blue
-
Components of Perls Prussian blue stain
-
Hemosiderosis at high magnification
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian