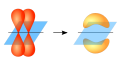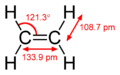Pi bond
Pi bond
A pi bond (Greek: π) is a type of chemical bond that is characterized by the sharing of electrons between atoms in a molecule through p orbitals that are in contact through two areas of overlap. Pi bonds are usually weaker than sigma bonds.
Etymology[edit]
The term "pi bond" is derived from the Greek letter π, which is used in mathematics to represent a constant. In the context of chemistry, the term "pi bond" is used to describe a type of covalent bond that is formed by the overlap of p orbitals.
Formation[edit]
Pi bonds are formed when two atomic orbitals come together to form a molecular orbital that is symmetrical along the bond axis. The p orbitals that participate in pi bond formation are in a parallel orientation and must be unhybridized. The electrons in pi bonds are sometimes referred to as "pi electrons".
Properties[edit]
Pi bonds are generally weaker than sigma bonds due to the poor overlap of p orbitals. However, they are crucial in the formation of double and triple bonds in molecules. Pi bonds also play a significant role in the structures of organic molecules, such as alkenes and alkynes.
Related Terms[edit]
See also[edit]
-
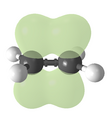
Ethylene 3D structure
-
Pi bond illustration
-
Ethane staggered conformation
-
Ethylene dimensions
-
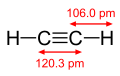
Acetylene dimensions
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian