Pseudotropine
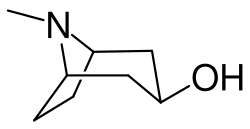
Pseudotropine is an alkaloid that is a structural isomer of tropine. It is a secondary metabolite found in a variety of plants, including those in the Solanaceae family. Pseudotropine is also known as isotropine and has been used in the synthesis of various pharmaceuticals.
Chemistry[edit]
Pseudotropine is a tropane alkaloid, a class of alkaloids that also includes atropine and cocaine. It is a bicyclic compound, consisting of a six-membered piperidine ring fused to a five-membered pyrrolidine ring. The compound is chiral, with the natural form being the (R)-enantiomer.
Biosynthesis[edit]
The biosynthesis of pseudotropine begins with the amino acids ornithine and arginine, which are converted into putrescine by the enzyme ornithine decarboxylase. Putrescine is then converted into N-methylputrescine by the enzyme putrescine N-methyltransferase. N-methylputrescine is then oxidized to form N-methyl-Δ1-pyrroline, which is then reduced to form tropinone. Tropinone is then rearranged to form pseudotropine by the enzyme tropinone reductase.
Pharmacology[edit]
Pseudotropine has been used as a starting material in the synthesis of various pharmaceuticals, including atropine, scopolamine, and hyoscyamine. These drugs are used to treat a variety of conditions, including gastrointestinal disorders, motion sickness, and certain eye diseases.
Toxicology[edit]
Like other tropane alkaloids, pseudotropine can be toxic if ingested in large amounts. Symptoms of overdose can include dry mouth, blurred vision, tachycardia, and hallucinations. In severe cases, overdose can lead to coma or death.
See also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
