Racemization
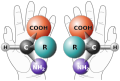
Racemization is a chemical process that converts an optically active compound into a racemic mixture, a 1:1 mixture of enantiomers. Enantiomers are pairs of molecules that are mirror images of each other but cannot be superimposed, much like one's left and right hands. This process is significant in various fields, including pharmaceuticals, biochemistry, and organic chemistry, due to its implications for the biological activity and properties of compounds.
Overview[edit]
In a racemic mixture, the physical properties such as boiling point, melting point, and density are identical for each enantiomer. However, their interaction with plane-polarized light and biological systems can be vastly different. One enantiomer may be therapeutically active, while the other is less active or may even produce adverse effects. This distinction is crucial in the development and use of pharmaceutical drugs.
Mechanism[edit]
Racemization can occur through several mechanisms, depending on the structure of the compound and the conditions it is subjected to. Common mechanisms include acid or base catalysis, where the protonation or deprotonation of a stereocenter leads to the formation of a planar intermediate, allowing for the reformation of the stereocenter in either configuration. Another mechanism involves the presence of a nucleophile or an electrophile that can attack the stereocenter, leading to its inversion.
Factors Influencing Racemization[edit]
Several factors can influence the rate of racemization, including temperature, pH, the presence of catalysts, and the structure of the compound itself. For example, higher temperatures generally increase the rate of racemization. The pH can affect the ionization state of the molecule, which in turn can influence its reactivity. The steric hindrance around the stereocenter can also affect the rate of racemization, with more hindered centers being less susceptible to racemization.
Applications[edit]
Racemization has important applications in the pharmaceutical industry, where the production of pure enantiomers of drug compounds is often desired due to their differing biological activities. Techniques such as chiral resolution and asymmetric synthesis are used to obtain enantiomerically pure compounds. Additionally, racemization is used in the field of amino acid dating, a technique used in paleontology and archeology to estimate the age of biological samples based on the racemization rates of amino acids.
Challenges[edit]
One of the main challenges associated with racemization is the potential loss of enantiomeric purity of pharmaceutical compounds, which can lead to reduced efficacy or increased side effects. Therefore, understanding and controlling racemization is crucial in the development and manufacturing of chiral drugs.
Conclusion[edit]
Racemization is a fundamental process in chemistry with significant implications for the pharmaceutical industry and beyond. Its understanding and control are essential for the development of effective and safe chiral drugs, as well as for applications in various scientific fields.
-
Chirality with hands
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian