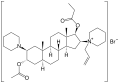
Rapacuronium bromide
Rapacuronium bromide is a neuromuscular-blocking drug or muscle relaxant in the category of non-depolarizing curare-like drugs, used in form of an injection to cause relaxation of the muscles during surgery. It was withdrawn from the market due to the risk of potentially fatal bronchospasm.
History[edit]
Rapacuronium bromide (INN, marketed as Raplon) was introduced in the late 1990s as a short-acting non-depolarizing muscle relaxant. It was developed by Organon International, a pharmaceutical company based in the Netherlands. However, it was withdrawn from the market in 2001 due to serious adverse effects.
Pharmacology[edit]
Rapacuronium bromide acts by competing for the cholinergic receptors at the motor end-plate. This action is antagonized by acetylcholinesterase inhibitors, such as neostigmine and edrophonium. Rapacuronium has a rapid onset and a short duration of action.
Adverse effects[edit]
The most serious adverse effect of rapacuronium bromide is bronchospasm. Other side effects include hypotension, tachycardia, and skin reactions at the injection site.
Withdrawal from the market[edit]
Rapacuronium bromide was withdrawn from the market in 2001 after reports of severe bronchospasm, including some that were fatal. The risk was particularly high in patients with a history of asthma.
See also[edit]
-
Rapacuronium bromide
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian