Sulfur

[[File:Nearly exhausted sulphur vat from which railroad cars are loaded, Freeport Sulphur Co., Hoskins Mound, Texas, 1a35438v.jpg|Nearly exhausted sulfur vat Sulfur or sulphur is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature.
Characteristics[edit]
Sulfur is a bright yellow crystalline solid. It is characterized by its odorous nature, often associated with the smell of rotten eggs, due to the presence of sulfur compounds. Sulfur is multivalent, meaning it has several common oxidation states, including -2, +2, +4 and +6.
Occurrence[edit]
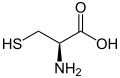
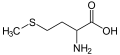
Sulfur occurs naturally as the pure element (native sulfur) and as sulfide and sulfate minerals. It is an essential element for life and is found in two amino acids, cysteine and methionine.
Uses[edit]
Sulfur is used in many industries, including the production of sulfuric acid, the world's most produced chemical. It is also used in the vulcanization of rubber, as a fungicide, and as a component in black gunpowder.
History[edit]
The history of sulfur is rooted in antiquity, with mentions of the element found in the Biblical texts. It was known to the ancients in its native form and was also part of the primitive gunpowder formulation.
Gallery =[edit]
-
Io
-
Sulfur mining
-
Kawah Ijen sulfur
-
Lapis lazuli block
-
Sulfur at carbon nanotube
-
L-Cysteine
-
Methionine
See also[edit]
External links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian