Thiosemicarbazone
Thiosemicarbazone[edit]
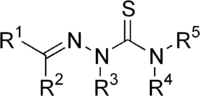
Thiosemicarbazones are a class of organic compounds characterized by the presence of the thiosemicarbazone functional group. This group is derived from thiosemicarbazide, which is a compound containing both a thioamide and a hydrazone moiety. Thiosemicarbazones have been studied for their potential applications in medicine, particularly as antiviral and anticancer agents.
Structure and Properties[edit]
Thiosemicarbazones are typically formed by the condensation of thiosemicarbazide with aldehydes or ketones. The general structure of a thiosemicarbazone can be represented as R1R2C=NNHC(S)NH2, where R1 and R2 are organic substituents that can vary widely, influencing the compound's properties and biological activity.
The presence of the thioamide group (C=S) and the hydrazone linkage (C=N-N) in thiosemicarbazones contributes to their ability to chelate metal ions, which is a key feature in their mechanism of action as therapeutic agents.
Biological Activity[edit]
Thiosemicarbazones have been extensively studied for their biological activities. They exhibit a wide range of pharmacological properties, including:
- Antiviral Activity: Some thiosemicarbazones have shown efficacy against viruses such as the influenza virus and the human immunodeficiency virus (HIV).
- Anticancer Activity: Thiosemicarbazones can inhibit the growth of cancer cells by interfering with metal ion homeostasis and inducing oxidative stress.
- Antibacterial and Antifungal Activity: These compounds have also demonstrated activity against various bacterial and fungal pathogens.
Mechanism of Action[edit]
The biological activity of thiosemicarbazones is often attributed to their ability to chelate metal ions such as iron and copper. By binding to these metal ions, thiosemicarbazones can disrupt essential biological processes in pathogens or cancer cells. For example, they can inhibit ribonucleotide reductase, an enzyme crucial for DNA synthesis, thereby preventing cell proliferation.
Synthesis[edit]
The synthesis of thiosemicarbazones typically involves the reaction of thiosemicarbazide with an aldehyde or ketone. This reaction forms a hydrazone linkage, resulting in the formation of the thiosemicarbazone compound. The reaction conditions can be adjusted to optimize yield and purity, and various substituents can be introduced to modify the biological activity of the resulting thiosemicarbazone.
Applications[edit]
Due to their diverse biological activities, thiosemicarbazones are being investigated for various therapeutic applications. They are considered promising candidates for the development of new antiviral and anticancer drugs. Research is ongoing to better understand their mechanisms of action and to develop derivatives with improved efficacy and reduced toxicity.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian