Threohydrobupropion
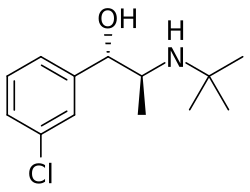
Threohydrobupropion is a metabolite of the antidepressant and smoking cessation aid bupropion. It is formed in the body through the reduction of bupropion by the enzyme carbonyl reductase. Threohydrobupropion, along with other metabolites such as hydroxybupropion and erythrohydrobupropion, contributes to the pharmacological effects of bupropion.
Pharmacology[edit]
Threohydrobupropion is known to inhibit the reuptake of the neurotransmitters dopamine and norepinephrine, similar to its parent compound bupropion. This inhibition increases the levels of these neurotransmitters in the brain, which is believed to contribute to its antidepressant and smoking cessation properties.
Metabolism[edit]
Bupropion is metabolized in the liver primarily by the enzyme cytochrome P450 2B6 (CYP2B6) to form hydroxybupropion. Threohydrobupropion is formed through the reduction of bupropion by carbonyl reductase. The pharmacokinetics of threohydrobupropion, including its half-life and bioavailability, are important for understanding the overall effects of bupropion therapy.
Clinical Significance[edit]
The presence of threohydrobupropion and its activity is significant in the clinical use of bupropion. The metabolite's ability to inhibit dopamine and norepinephrine reuptake supports the therapeutic effects of bupropion in treating depression and aiding in smoking cessation. Understanding the role of threohydrobupropion can help in optimizing bupropion dosing and managing potential side effects.
See Also[edit]
References[edit]
-
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian