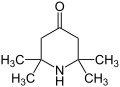
Triacetonamine
Chemical compound
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Triacetonamine is an organic compound with the formula C9H17NO. It is a cyclic amine that is derived from acetone.
Structure and properties[edit]
Triacetonamine is a cyclic compound that features a six-membered ring containing a nitrogen atom. The compound is formed by the condensation of three molecules of acetone with one molecule of ammonia. This reaction results in the formation of a stable, crystalline solid.
Synthesis[edit]
The synthesis of triacetonamine involves the reaction of acetone with ammonia. The process typically requires a catalyst and is conducted under controlled conditions to ensure the formation of the desired product. The reaction can be represented as follows:
3 (CH3)2CO + NH3 _ C9H17NO + 3 H2O
Applications[edit]
Triacetonamine is used in various chemical applications, primarily as an intermediate in the synthesis of other compounds. It serves as a building block in the production of pharmaceuticals, agrochemicals, and other specialized chemicals.
Safety and handling[edit]
As with many chemical compounds, proper safety precautions should be taken when handling triacetonamine. It is important to use appropriate personal protective equipment and to follow standard safety protocols to prevent exposure.
Related pages[edit]
References[edit]
-
Structural formula of Triacetonamine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian