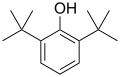
2,6-Di-tert-butylphenol
2,6-Di-tert-butylphenol[edit]

2,6-Di-tert-butylphenol is an organic compound with the formula C14H22O. It is a type of phenol, which is a class of aromatic compounds characterized by a hydroxyl group (-OH) attached to a benzene ring.
Structure and Properties[edit]
2,6-Di-tert-butylphenol is a substituted phenol where two tert-butyl groups are attached to the benzene ring at the 2 and 6 positions. This substitution pattern provides the compound with significant steric hindrance, which affects its chemical reactivity and physical properties.
The presence of the bulky tert-butyl groups makes 2,6-Di-tert-butylphenol a hindered phenol, which is often used as an antioxidant in various applications. The compound is a white crystalline solid at room temperature.
Applications[edit]
2,6-Di-tert-butylphenol is primarily used as an antioxidant. Antioxidants are substances that can prevent or slow damage to cells caused by free radicals, which are unstable molecules that the body produces as a reaction to environmental and other pressures.
In industrial applications, 2,6-Di-tert-butylphenol is used to stabilize polymers and plastics, preventing degradation due to oxidation. It is also used in lubricants and fuels to enhance their stability and performance.
Synthesis[edit]
The synthesis of 2,6-Di-tert-butylphenol typically involves the alkylation of phenol with isobutylene in the presence of an acid catalyst. This process results in the selective formation of the 2,6-di-tert-butyl derivative due to the steric and electronic effects that direct the alkylation to these positions on the phenol ring.
Safety and Environmental Impact[edit]
As with many phenolic compounds, 2,6-Di-tert-butylphenol should be handled with care. It can cause skin and eye irritation upon contact. Proper safety precautions should be taken when handling this chemical in industrial or laboratory settings.
The environmental impact of 2,6-Di-tert-butylphenol is a subject of study, as it can persist in the environment and may have effects on aquatic life. Efforts are made to minimize its release into the environment and to develop more biodegradable alternatives.
Related pages[edit]
2,6-Di-tert-butylphenol[edit]
-
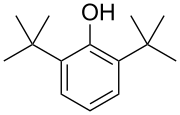
2D structure of 2,6-Di-tert-butylphenol
-
3D ball-and-stick model of 2,6-Di-tert-butylphenol
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian