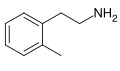
2-Methylphenethylamine
2-Methylphenethylamine (also known as 2MPEA) is a stimulant of the phenethylamine class. It is a positional isomer of amphetamine and is used in the manufacture of certain pharmaceuticals.
Chemistry[edit]
2-Methylphenethylamine is a phenethylamine, which is a group of organic compounds that include a phenyl ring directly attached to an amino group. It is a positional isomer of amphetamine, meaning it has the same molecular formula but a different arrangement of atoms.
Pharmacology[edit]
As a phenethylamine, 2-Methylphenethylamine has similar effects to other stimulants in this class. It increases levels of dopamine, norepinephrine, and serotonin in the brain, leading to increased arousal, alertness, and mood elevation.
Uses[edit]
2-Methylphenethylamine is used in the manufacture of certain pharmaceuticals. It is also used as a starting material in the synthesis of various psychoactive substances.
Safety[edit]
The safety profile of 2-Methylphenethylamine is not well-studied. As a stimulant, it may have potential for abuse and addiction. It may also have side effects similar to other stimulants, such as increased heart rate, blood pressure, and risk of stroke.
See also[edit]
-
2-Methylphenethylamine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian