2C-H
2C-H[edit]
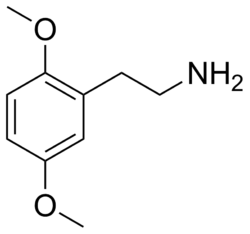
2C-H (2,5-dimethoxyphenethylamine) is a psychedelic phenethylamine of the 2C family. It is the base compound for a series of psychedelic phenethylamines, which include 2C-B, 2C-I, and 2C-E.
2C-H is a synthetic compound that was first synthesized by Alexander Shulgin. It is known for its role as a precursor in the synthesis of other 2C compounds, but it is not commonly used as a recreational drug due to its relatively mild effects compared to its derivatives.
Chemical Properties[edit]
2C-H is a phenethylamine with two methoxy groups attached to the benzene ring at the 2 and 5 positions. Its chemical formula is C10H15NO2. The presence of the methoxy groups is a defining feature of the 2C family of compounds.
Pharmacology[edit]
2C-H acts primarily as a serotonin receptor agonist, similar to other compounds in the 2C series. However, its affinity for these receptors is lower than that of its more potent derivatives, such as 2C-B and 2C-I. This results in a less intense psychedelic experience.
Synthesis[edit]
2C-H can be synthesized from 2,5-dimethoxybenzaldehyde through a series of chemical reactions. The synthesis typically involves the formation of a nitrostyrene intermediate, which is then reduced to the final phenethylamine product.
Legal Status[edit]
The legal status of 2C-H varies by country. In some jurisdictions, it is classified as a controlled substance due to its structural similarity to other psychoactive compounds. In others, it may be legal or unregulated.
Related Compounds[edit]
2C-H is the parent compound of the 2C series, which includes a variety of other phenethylamines with different substituents on the benzene ring. Some notable derivatives include:
These compounds are known for their psychedelic effects and have been used in both recreational and research settings.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian