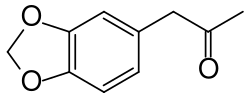
3,4-Methylenedioxyphenylpropan-2-one
Chemical compound
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
3,4-Methylenedioxyphenylpropan-2-one (MDP2P), also known as piperonyl methyl ketone (PMK), is an organic compound with the molecular formula C10H10O3. It is a key intermediate in the synthesis of the psychoactive drug MDMA (3,4-methylenedioxy-N-methylamphetamine), commonly known as ecstasy.
Chemical Properties[edit]
3,4-Methylenedioxyphenylpropan-2-one is a colorless to pale yellow liquid with a density of 1.205 g/cm³. It has a melting point of 7.5 °C and a boiling point of 290 °C. The compound is characterized by the presence of a methylenedioxy group attached to a phenyl ring, which is further connected to a propan-2-one moiety.
Synthesis[edit]
MDP2P can be synthesized through several methods, including the Wacker oxidation of safrole or isomerization of isosafrole. These methods are often employed in clandestine laboratories due to the compound's role as a precursor in the illicit production of MDMA.
Legal Status[edit]
Due to its use in the synthesis of MDMA, 3,4-methylenedioxyphenylpropan-2-one is classified as a List I chemical under the United States Controlled Substances Act. This classification imposes strict regulatory controls on its manufacture, distribution, and possession.
Applications[edit]
While its primary notoriety comes from its role in the synthesis of MDMA, MDP2P also has legitimate applications in the synthesis of other chemicals and pharmaceuticals. However, its legal status significantly restricts its availability and use.
Safety and Handling[edit]
MDP2P is classified as a hazardous substance and should be handled with care. It has a flash point of 127 °C, indicating that it can form flammable vapors at relatively low temperatures. Appropriate safety measures, including the use of personal protective equipment and proper ventilation, should be employed when handling this compound.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian