4-Dehydroepiandrosterone
A steroid hormone
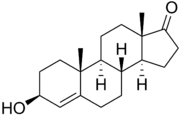
4-Dehydroepiandrosterone (4-DHEA) is a steroid hormone that is a naturally occurring androgen and estrogen precursor. It is closely related to dehydroepiandrosterone (DHEA), differing only by the position of a double bond in the steroid structure.
Biochemistry[edit]
4-DHEA is synthesized in the adrenal glands, gonads, and the brain. It serves as a precursor to testosterone and estradiol, which are critical for the development and maintenance of male and female secondary sexual characteristics, respectively. The conversion of 4-DHEA to these active hormones involves several enzymatic steps, primarily occurring in peripheral tissues.
Functions[edit]
4-DHEA plays a role in various physiological processes, including:
- Immune system modulation
- Metabolism regulation
- Bone density maintenance
- Mood and cognitive function enhancement
Clinical Significance[edit]
4-DHEA has been studied for its potential therapeutic applications in conditions such as adrenal insufficiency, depression, and osteoporosis. However, its use as a supplement is controversial due to potential side effects and the lack of comprehensive clinical trials.
Regulation[edit]
In many countries, 4-DHEA is regulated as a controlled substance due to its potential for misuse in performance enhancement and bodybuilding. It is important to consult with healthcare professionals before considering its use.
See also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian