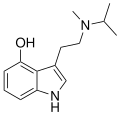
4-HO-MiPT
4-HO-MiPT
A psychedelic compound
4-HO-MiPT (4-hydroxy-N-methyl-N-isopropyltryptamine) is a psychedelic compound of the tryptamine class. It is structurally related to psilocin, the active component in psilocybin mushrooms.
Chemical structure and properties[edit]
4-HO-MiPT is a synthetic compound that belongs to the tryptamine family. Its chemical structure is characterized by a tryptamine backbone with a hydroxy group at the 4-position and an N-methyl-N-isopropyl substitution on the amine nitrogen. This structure is similar to that of psilocin, differing only in the substitution pattern on the nitrogen atom.
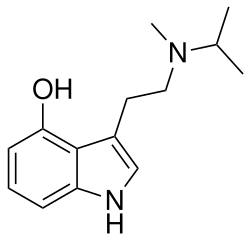
The molecular formula of 4-HO-MiPT is C14H20N2O, and it has a molecular weight of 232.32 g/mol. It is typically encountered as a white or off-white crystalline powder.
Pharmacology[edit]
4-HO-MiPT acts primarily as a serotonin receptor agonist, with a high affinity for the 5-HT2A receptor. This action is thought to be responsible for its psychedelic effects. The compound is known to produce alterations in perception, mood, and cognition, similar to other psychedelics such as LSD and psilocybin.
Effects[edit]
The effects of 4-HO-MiPT are dose-dependent and can vary significantly between individuals. Common effects include visual and auditory hallucinations, changes in thought patterns, and an altered sense of time. Users may also experience euphoria, introspection, and spiritual experiences. The onset of effects typically occurs within 20 to 40 minutes after ingestion, with the peak lasting 2 to 4 hours and a total duration of 4 to 6 hours.
Safety and legality[edit]
The safety profile of 4-HO-MiPT is not well-established, as it has not been extensively studied in humans. As with other psychedelics, there is a potential for psychological distress, including anxiety and paranoia, particularly at higher doses. The legal status of 4-HO-MiPT varies by country, and it may be classified as a controlled substance in some jurisdictions.
Synthesis[edit]
4-HO-MiPT can be synthesized from tryptamine through a series of chemical reactions, including methylation and isopropylation of the amine group, followed by hydroxylation at the 4-position. The synthesis requires specialized knowledge in organic chemistry and access to appropriate laboratory equipment.
Related compounds[edit]
4-HO-MiPT is part of a family of substituted tryptamines, which includes other compounds such as 4-AcO-MiPT, 4-HO-DMT, and 4-HO-DET. These compounds share similar structures and pharmacological properties, but differ in their potency and duration of effects.
Related pages[edit]
Gallery[edit]
-
3D stick model of 4-HO-MiPT
4-HO-MiPT[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian