9-Carboxymethoxymethylguanine
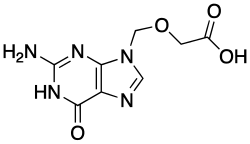
9-Carboxymethoxymethylguanine (CMMG) is a nucleoside analogue and a metabolite of the antiviral drug acyclovir. It is primarily known for its role in the treatment of herpes simplex virus (HSV) infections.
Chemical Structure and Properties[edit]
9-Carboxymethoxymethylguanine is a derivative of guanine, one of the four main nucleobases found in DNA and RNA. The chemical structure of CMMG includes a carboxymethoxymethyl group attached to the ninth position of the guanine base. This modification is crucial for its antiviral activity.
Mechanism of Action[edit]
CMMG, like its parent compound acyclovir, works by inhibiting the replication of viral DNA. It is incorporated into the viral DNA chain during replication, leading to premature chain termination. This action effectively halts the proliferation of the virus within the host cells.
Pharmacokinetics[edit]
After administration of acyclovir, CMMG is formed as a metabolite through the action of cellular enzymes. The pharmacokinetics of CMMG involve its absorption, distribution, metabolism, and excretion. It is primarily excreted through the renal system.
Clinical Uses[edit]
CMMG is not used directly as a therapeutic agent but is an important metabolite in the pharmacological activity of acyclovir. Acyclovir is widely used in the treatment of herpes simplex virus infections, including herpes labialis, genital herpes, and herpes zoster.
Side Effects and Toxicity[edit]
The side effects of CMMG are generally related to its parent compound, acyclovir. Common side effects include nausea, vomiting, and diarrhea. In rare cases, high levels of CMMG can lead to neurotoxicity, manifesting as confusion, hallucinations, and seizures.
Research and Development[edit]
Ongoing research is focused on understanding the detailed mechanisms of CMMG and its potential applications in antiviral therapy. Studies are also investigating the development of new nucleoside analogues with improved efficacy and reduced toxicity.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian