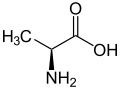
Alanine
Alanine

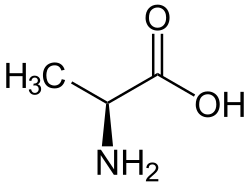
Alanine is an alpha-amino acid that is used in the biosynthesis of proteins. It contains an amino group, a carboxylic acid group, and a side chain methyl group, making it a non-polar, aliphatic amino acid. Alanine is classified as a non-essential amino acid, meaning it can be synthesized by the human body and does not need to be obtained directly through the diet.
Structure[edit]
Alanine has the chemical formula C3H7NO2. The IUPAC name for alanine is 2-aminopropanoic acid. The molecular structure of alanine consists of a central carbon atom (the alpha carbon) bonded to an amino group (NH2), a carboxyl group (COOH), a hydrogen atom, and a methyl group (CH3), which is the side chain.
Stereochemistry[edit]
Alanine exists in two enantiomers, L-alanine and D-alanine, which are mirror images of each other. In biological systems, L-alanine is the form that is incorporated into proteins. The chirality of alanine is important for its function in protein structure and enzyme activity.
Biosynthesis[edit]
Alanine can be synthesized in the body through the transamination of pyruvate, a key intermediate in glycolysis. This reaction involves the transfer of an amino group from an amino acid, such as glutamate, to pyruvate, forming alanine and alpha-ketoglutarate.
Function[edit]
Alanine plays a crucial role in glucose-alanine cycle, which helps in the transport of nitrogen from muscle tissue to the liver. In this cycle, alanine is formed in muscle cells by the transamination of pyruvate. It is then transported to the liver, where it is converted back to pyruvate, which can be used for gluconeogenesis to produce glucose.
Metabolism[edit]
In the liver, alanine can be converted into pyruvate by the enzyme alanine transaminase. This pyruvate can then enter the citric acid cycle or be used for gluconeogenesis. Alanine is also involved in the synthesis of other amino acids and metabolic pathways.
Zwitterion Form[edit]
In aqueous solution, alanine exists predominantly as a zwitterion, a molecule with both positive and negative charges. The amino group is protonated (NH3+), and the carboxyl group is deprotonated (COO-), resulting in a neutral overall charge.
Gallery[edit]
-
Synthesis of alanine - step 1
-
Synthesis of alanine - step 2
-
Zwitterionic form of alanine
Related pages[edit]
-
Alanine
-
L-Alanine structure
-
Alanine
-
Synthesis of alanine - step 1
-
Synthesis of alanine - step 2
-
Zwitterion form of alanine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian