Aldose reductase
Aldose reductase
Aldose reductase is an enzyme that plays a crucial role in the polyol pathway, which is involved in the metabolism of glucose. It is a member of the aldo-keto reductase superfamily and is encoded by the AKR1B1 gene in humans. This enzyme catalyzes the reduction of glucose to sorbitol, using NADPH as a cofactor.
Structure[edit]
Aldose reductase is a monomeric protein with a molecular weight of approximately 36 kDa. The enzyme's active site is characterized by a conserved NADPH binding domain, which is essential for its catalytic activity. The structure of aldose reductase has been elucidated through X-ray crystallography, revealing a typical (_/_)8-barrel fold.
Function[edit]
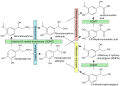
The primary function of aldose reductase is to catalyze the conversion of glucose to sorbitol in the polyol pathway. This reaction is the first step in the pathway and is followed by the oxidation of sorbitol to fructose by sorbitol dehydrogenase. The polyol pathway is particularly active in tissues where glucose uptake is insulin-independent, such as the lens of the eye, peripheral nerves, and the kidney.
Clinical Significance[edit]
Aldose reductase has been implicated in the development of diabetic complications, such as diabetic retinopathy, diabetic neuropathy, and diabetic nephropathy. In conditions of hyperglycemia, excess glucose is shunted through the polyol pathway, leading to the accumulation of sorbitol and fructose. This can result in osmotic and oxidative stress, contributing to tissue damage.
Inhibition[edit]
Aldose reductase inhibitors (ARIs) are a class of drugs that have been developed to prevent or reduce the complications of diabetes by inhibiting the activity of aldose reductase. These inhibitors aim to reduce the flux of glucose through the polyol pathway, thereby decreasing the accumulation of sorbitol and its associated toxic effects.
Mechanism[edit]
The catalytic mechanism of aldose reductase involves the transfer of a hydride ion from NADPH to the carbonyl group of glucose, reducing it to sorbitol. The enzyme's active site facilitates this reaction through a series of hydrogen bonds and hydrophobic interactions that stabilize the transition state.
Related pages[edit]
Gallery[edit]
-
Structure of aldose reductase
-
Polyol pathway
-
Mechanism of aldose reductase
-
NADPH binding in aldose reductase
-
Noradrenaline breakdown
Aldose_reductase[edit]
-
Aldose reductase structure
-
Polyol pathway
-
Final mechanism of aldose reductase
-
NADPH hydrogen bonded
-
Noradrenaline breakdown
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian