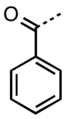
Benzoyl group
Benzoyl group refers to a functional group with the formula C6H5CO-, derived from benzoic acid. It consists of a phenyl group (C6H5-) attached to a carbonyl group (CO-). This group is notable for its application in various chemical reactions and its presence in numerous pharmaceuticals and organic compounds.
Structure and Properties[edit]
The benzoyl group is characterized by its aromatic ring, which contributes to its chemical stability and reactivity. The presence of the carbonyl group enhances its reactivity, making it a key functional group in organic synthesis. The electron-withdrawing nature of the carbonyl group affects the electron density of the aromatic ring, influencing its chemical behavior.
Synthesis[edit]
Benzoyl groups can be introduced into organic compounds through several methods. One common approach is the Friedel-Crafts acylation, where an aromatic compound reacts with an acyl chloride in the presence of a Lewis acid catalyst, such as AlCl3. Another method involves the oxidation of benzyl compounds or the use of benzoyl chloride in a reaction with alcohols or amines to form esters or amides, respectively.
Applications[edit]
Benzoyl groups are prevalent in various chemical and pharmaceutical applications. They are used in the synthesis of polymers, dyes, and fragrances, as well as in the preparation of pesticides and pharmaceuticals. One well-known compound containing a benzoyl group is benzoyl peroxide, widely used in acne treatment due to its antimicrobial properties and ability to promote skin peeling.
Reactivity[edit]
The reactivity of the benzoyl group is influenced by the aromatic system and the carbonyl functionality. It undergoes typical carbonyl reactions, such as nucleophilic addition and condensation reactions. The aromatic ring can participate in electrophilic aromatic substitution, although the presence of the carbonyl group can deactivate the ring towards further substitution.
Safety and Environmental Impact[edit]
Compounds containing benzoyl groups can vary in their safety and environmental impact. Benzoyl peroxide, for example, is generally considered safe for topical use but can cause skin irritation in some individuals. Environmental considerations depend on the specific compound and its degradation products.
-
Benzoyl Radical
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian