C-Raf
Protein-coding gene in the species Homo sapiens
| Symbol | RAF1 |
|---|---|
| HGNC ID | 9829 |
| Alternative symbols | – |
| Entrez Gene | – |
| OMIM | 164760 |
| RefSeq | NM_002880 |
| UniProt | P04049 |
| Chromosome | 3p25 |
| Locus supplementary data | – |
C-Raf (also known as RAF1) is a protein kinase that is encoded by the RAF1 gene in Homo sapiens. It is a member of the Raf kinase family of serine/threonine-specific protein kinases.
Function[edit]
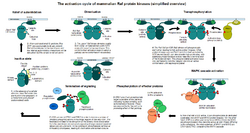
C-Raf plays a crucial role in the MAPK/ERK pathway, which is involved in the regulation of cell division, differentiation, and secretion. Upon activation by Ras proteins, C-Raf phosphorylates and activates MEK1 and MEK2, which in turn activate ERK1 and ERK2. This signaling cascade is essential for transmitting signals from the cell membrane to the nucleus.
Structure[edit]
C-Raf consists of three conserved regions: CR1, CR2, and CR3. The CR1 region contains the Ras-binding domain (RBD) and the cysteine-rich domain (CRD), which are essential for its interaction with Ras proteins. The CR2 region is a serine/threonine-rich domain that serves as a regulatory region. The CR3 region contains the kinase domain, which is responsible for its enzymatic activity.
Clinical Significance[edit]
Mutations in the RAF1 gene have been associated with several types of cancer, including melanoma, lung cancer, and colorectal cancer. Additionally, mutations in C-Raf are linked to developmental disorders such as Noonan syndrome and LEOPARD syndrome.
Interactions[edit]
C-Raf interacts with several proteins, including:
Research[edit]
Ongoing research is focused on understanding the precise mechanisms of C-Raf activation and its role in various diseases. Inhibitors targeting C-Raf and other components of the MAPK/ERK pathway are being developed as potential therapeutic agents for cancer treatment.
See also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian