Calcium oxalate
Calcium Oxalate[edit]

Calcium oxalate is a chemical compound with the formula CaC₂O₄. It is a salt of calcium and oxalic acid, and it occurs naturally in many plants and animals. Calcium oxalate is known for its role in the formation of kidney stones and is also found in certain foods and plants.
Structure and Properties[edit]
Calcium oxalate exists in several hydrate forms, including the monohydrate, dihydrate, and trihydrate. The monohydrate form, known as whewellite, is the most common form found in kidney stones. The dihydrate form is known as weddellite, and the trihydrate form is called caoxite.
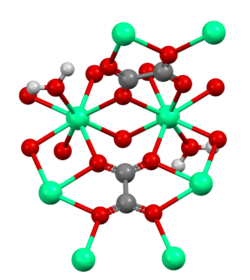
The compound is poorly soluble in water, which contributes to its tendency to precipitate and form stones in the urinary tract. Calcium oxalate crystals can appear in various shapes, including needle-like, dumbbell-shaped, or envelope-shaped, depending on the conditions of formation.
Occurrence[edit]
Calcium oxalate is found in many plants, where it serves as a defense mechanism against herbivory. Plants such as rhubarb, spinach, and beetroot contain high levels of oxalates. In animals, calcium oxalate is a common component of kidney stones, which can cause significant health issues if not managed properly.

Role in Kidney Stones[edit]
Calcium oxalate is the most common constituent of kidney stones, accounting for about 80% of cases. Stones form when there is an imbalance in the concentration of calcium and oxalate in the urine, leading to precipitation and crystallization. Factors such as dehydration, dietary habits, and genetic predisposition can influence stone formation.
Detection and Analysis[edit]
Calcium oxalate crystals can be detected in urine samples using microscopy. They are often identified by their characteristic shapes and birefringence under polarized light.
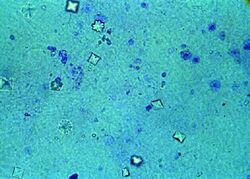
Prevention and Treatment[edit]
Preventing calcium oxalate stone formation involves maintaining adequate hydration, reducing dietary oxalate intake, and managing calcium levels. In some cases, medications may be prescribed to alter urine chemistry and reduce stone formation risk.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian