Carbylamine reaction
Carbylamine Reaction[edit]
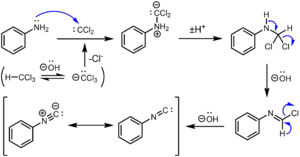
The carbylamine reaction, also known as the Hofmann isocyanide synthesis, is a chemical reaction that involves the synthesis of isocyanides from primary amines, chloroform, and a base. This reaction is a classic method for the preparation of isocyanides, which are compounds characterized by the functional group -N_C.
Mechanism[edit]
The mechanism of the carbylamine reaction involves several steps:
- The base, typically potassium hydroxide (KOH), deprotonates the chloroform to form the trichloromethanide ion.
- The trichloromethanide ion undergoes an _-elimination to generate dichlorocarbene (:CCl_).
- The dichlorocarbene then reacts with the primary amine to form an intermediate N-chloromethylamine.
- A second deprotonation occurs, leading to the formation of an isocyanide and the release of two molecules of hydrochloric acid.
The overall reaction can be summarized as follows:
- RNH_ + CHCl_ + 3KOH _ RNC + 3KCl + 3H_O
Applications[edit]
The carbylamine reaction is primarily used in organic synthesis to prepare isocyanides, which are valuable intermediates in the synthesis of various heterocyclic compounds and pharmaceuticals. Isocyanides are also used in the Ugi reaction, a multicomponent reaction that is important in combinatorial chemistry.
Limitations[edit]
The carbylamine reaction is specific to primary amines. Secondary and tertiary amines do not undergo this reaction. Additionally, the reaction is known for its unpleasant odor due to the formation of isocyanides, which are notoriously malodorous.
Safety[edit]
Due to the production of isocyanides, which are toxic and have a strong odor, the carbylamine reaction should be conducted in a well-ventilated area or under a fume hood. Proper personal protective equipment (PPE) such as gloves and goggles should be worn.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian