Chemical bond
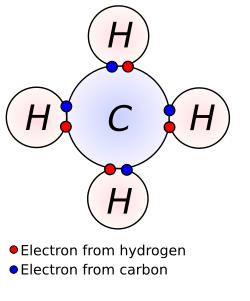

Chemical bond refers to the lasting attraction between atoms, ions, or molecules that enables the formation of chemical compounds. The strength of chemical bonds varies considerably; there are "strong bonds" such as covalent bonds and ionic bonds that constitute the primary structure of substances, and "weak bonds" such as hydrogen bonds and van der Waals forces that play crucial roles in the structure and properties of molecules.
Types of Chemical Bonds[edit]
The main types of chemical bonds include:
Ionic Bonds[edit]
An ionic bond is formed when one atom transfers an electron to another atom, resulting in the formation of positively charged cations and negatively charged anions. This type of bond typically occurs between metals and non-metals. Sodium chloride (NaCl) is a classic example of an ionic compound.
Covalent Bonds[edit]
A covalent bond is formed when two atoms share one or more pairs of electrons. This type of bond is characterized by the formation of molecules and can be found in both organic and inorganic compounds. The number of electron pairs shared between atoms can vary, leading to single, double, or triple covalent bonds.
Metallic Bonds[edit]
Metallic bonds are formed between atoms of metallic elements. In these bonds, electrons are not shared or transferred between atoms. Instead, they are delocalized and move freely throughout the entire structure, which explains the conductivity, malleability, and ductility of metals.
Hydrogen Bonds[edit]
A hydrogen bond is a weak bond that occurs when a hydrogen atom covalently bonded to a highly electronegative atom, such as oxygen or nitrogen, experiences attraction to another electronegative atom. Hydrogen bonds are crucial in the structure of DNA and proteins, as well as in the properties of water.
Van der Waals Forces[edit]
Van der Waals forces are weak intermolecular forces that include attractions and repulsions between atoms, molecules, and surfaces, as well as other intermolecular forces. They are named after Dutch physicist Johannes Diderik van der Waals.
Chemical Bonding Theories[edit]
Several theories have been developed to explain the formation and behavior of chemical bonds, including:
- Valence Bond Theory: This theory explains how electrons in atoms create chemical bonds by overlapping atomic orbitals to form covalent bonds.
- Molecular Orbital Theory: This theory describes how electrons are delocalized within a molecule and occupy molecular orbitals that can extend over the entire molecule.
- Lewis Theory: Introduced by Gilbert N. Lewis, it focuses on the role of valence electrons in bond formation and the concept of electron dot structures.
Importance of Chemical Bonds[edit]
Chemical bonds are fundamental to the study of chemistry. They explain the structure, properties, and reactivity of all chemical substances. Understanding chemical bonds allows scientists to predict the behavior of compounds, design new materials, and understand biological processes at a molecular level.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
