Chemical formula

Chemical formula represents a way of expressing information about the proportions of atoms that constitute a particular chemical compound, using a single line of chemical element symbols and numbers. It is a fundamental concept in the field of chemistry, serving as a concise means for conveying complex information about chemical substances.
Types of Chemical Formulas[edit]
There are several types of chemical formulas, each serving a different purpose and providing different levels of detail about the chemical composition and structure of a compound.
Empirical Formula[edit]
The empirical formula of a chemical compound is the simplest positive integer ratio of atoms present in a compound. It does not provide information about the exact number of atoms or the arrangement of atoms in a molecule but gives the simplest ratio of the elements. For example, the empirical formula of glucose is CH2O, indicating that the elements carbon (C), hydrogen (H), and oxygen (O) are present in a 1:2:1 ratio.
Molecular Formula[edit]
The molecular formula provides the exact number of each type of atom in a molecule, but like the empirical formula, it does not convey information about the arrangement of those atoms. The molecular formula of glucose is C6H12O6, indicating that a molecule of glucose contains 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms.
Structural Formula[edit]
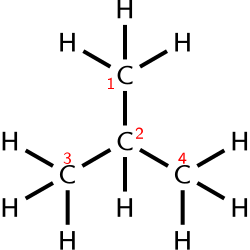
The structural formula goes a step further by showing the arrangement of atoms within a molecule. It can be represented in various ways, including the Lewis structure, which uses dots and lines to represent electrons and bonds, respectively. Structural formulas are crucial for understanding the chemical behavior of compounds.
Condensed Formula[edit]
A condensed formula is a more compact form of the structural formula. It shows the arrangement of atoms in a way that highlights the molecular structure but in a less detailed manner than a full structural formula. For example, the condensed formula for acetic acid might be written as CH3COOH, indicating the connections between atoms.
Importance of Chemical Formulas[edit]
Chemical formulas are essential in the field of chemistry for several reasons: - They provide a standard way of describing chemical substances. - They allow chemists to easily communicate complex information about chemical compositions and reactions. - They are crucial for calculating the stoichiometry of chemical reactions, which involves measuring the quantities of reactants and products. - They help in identifying and understanding the properties of new compounds.
Reading Chemical Formulas[edit]
Understanding chemical formulas requires knowledge of the symbols for chemical elements, which are standardized and internationally recognized. The number of atoms of an element in a compound is indicated by a subscript number following the element symbol. If no subscript is present, it is assumed that there is only one atom of that element in the compound.
Conclusion[edit]
Chemical formulas are a fundamental aspect of chemistry, providing a concise and informative way to describe the composition of chemical compounds. They play a crucial role in the study and application of chemistry, from the classroom to advanced research in the development of new materials and medicines.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
