Stoichiometry
Stoichiometry[edit]
Stoichiometry is a branch of chemistry that deals with the quantitative relationships between the amounts of reactants and products in a chemical reaction. It is based on the law of conservation of mass, which states that matter is neither created nor destroyed in a chemical reaction. Stoichiometry allows chemists to predict the quantities of substances consumed and produced in a given reaction.
Basic Concepts[edit]
Stoichiometry involves several key concepts, including:
Mole Concept[edit]
The mole is a fundamental unit in chemistry that represents a specific number of particles, usually atoms or molecules. One mole is defined as exactly 6.022 x 10^23 particles, known as Avogadro's number. The mole allows chemists to convert between the mass of a substance and the number of particles it contains.
Balanced Chemical Equations[edit]
A chemical equation represents a chemical reaction using symbols and formulas. A balanced chemical equation has the same number of each type of atom on both sides of the equation, reflecting the conservation of mass. Balancing chemical equations is a crucial step in stoichiometry.
Molar Ratios[edit]
Molar ratios are derived from the coefficients of a balanced chemical equation. They indicate the proportions of reactants and products involved in the reaction. These ratios are used to calculate the amounts of substances consumed or produced.
Stoichiometric Calculations[edit]
Stoichiometric calculations involve using the balanced chemical equation to determine the quantities of reactants and products. The steps typically include:
1. Writing and Balancing the Chemical Equation: Ensure the chemical equation is balanced. 2. Converting Mass to Moles: Use the molar mass of substances to convert between mass and moles. 3. Using Molar Ratios: Apply the molar ratios from the balanced equation to find the moles of other substances. 4. Converting Moles to Mass: Convert moles back to mass if needed.
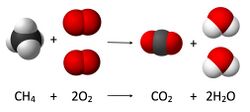
Example: Combustion of Methane[edit]
The combustion of methane is a common example used to illustrate stoichiometry. The balanced chemical equation for the combustion of methane is:
\[ \text{CH}_4 + 2\text{O}_2 \rightarrow \text{CO}_2 + 2\text{H}_2\text{O} \]
In this reaction, one mole of methane reacts with two moles of oxygen to produce one mole of carbon dioxide and two moles of water. Using stoichiometry, one can calculate the amount of oxygen needed to completely combust a given amount of methane, or the amount of carbon dioxide produced.
Related Pages[edit]
| Branches of chemistry | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian