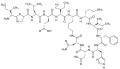
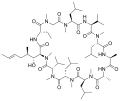
Cyclic peptide
Cyclic peptides are a class of peptides in which the amino acid sequence forms a ring structure rather than a linear chain. These peptides are produced by a variety of organisms including bacteria, plants, and animals, and have attracted interest due to their diverse range of biological activities and their potential applications in medicine, biotechnology, and material science.
Structure and Biosynthesis[edit]
Cyclic peptides are characterized by their cyclic structure, which is formed through a covalent bond between the N-terminus and C-terminus of the peptide chain or through internal side-chain-to-side-chain bonds. This cyclic structure imparts greater stability against enzymatic degradation compared to their linear counterparts, making them more resilient in biological environments.
Biosynthetically, cyclic peptides can be generated through nonribosomal peptide synthetases (NRPS), ribosomally synthesized and post-translationally modified peptides (RiPPs), or through chemical synthesis. NRPS are large, multi-enzyme complexes that assemble cyclic peptides in a template-independent manner, allowing for the incorporation of non-proteinogenic amino acids. RiPPs are initially synthesized as linear precursors by the ribosome and subsequently cyclized by specific enzymes.
Biological Activities and Applications[edit]
Cyclic peptides exhibit a wide range of biological activities, including antimicrobial, antitumor, immunosuppressive, and hormone-like activities. Their ability to mimic protein surfaces makes them excellent candidates for disrupting protein-protein interactions, which are often undruggable by small molecules and too complex for traditional biologics.
In medicine, cyclic peptides are being explored as therapeutic agents due to their high specificity and affinity for targets, combined with their relative stability and low toxicity. Examples include the antimicrobial peptide polymyxin B, the immunosuppressant cyclosporine, and the tumor-homing peptide cycloRGDfV.
In biotechnology, cyclic peptides are used as tools for drug discovery and as biosensors. Their modifiable structure allows for the design of molecules with tailored properties, such as increased stability or altered specificity.
Challenges and Future Directions[edit]
Despite their potential, the development of cyclic peptides as therapeutic agents faces several challenges. These include difficulties in predicting their structure-activity relationships, challenges in oral bioavailability due to their size and physicochemical properties, and the complexity of their synthesis and modification.
Advances in computational methods for the design and optimization of cyclic peptides, combined with innovative approaches to their synthesis and delivery, are expected to overcome these challenges. The ongoing discovery of naturally occurring cyclic peptides and their mechanisms of action also continues to provide valuable insights into their potential applications.
See Also[edit]
This peptide related article is a stub. You can help WikiMD by expanding it.
-
Alpha-amanitin structure
-
Bacitracin
-
Ciclosporin
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian