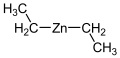Diethylzinc
Diethylzinc (IUPAC name: diethylzinc) is an organozinc compound with the formula Zn(C2H5)2. It is a highly reactive, colorless liquid which is sensitive to air and moisture, making it a challenging substance to work with under standard laboratory conditions. Diethylzinc is used primarily as a reagent in the synthesis of organic compounds and as a precursor to other zinc compounds.
Properties[edit]
Diethylzinc is a volatile, pyrophoric liquid that ignites spontaneously in air. It is soluble in hydrocarbons, ethers, and other organic solvents, but reacts violently with water, alcohols, and acids to produce ethane and zinc salts. The compound has a boiling point of 118°C and a melting point of -28°C.
Synthesis[edit]
Diethylzinc can be synthesized by the reaction of ethyl iodide with zinc metal. This process involves the direct reaction of zinc with ethyl iodide in the presence of anhydrous ether or tetrahydrofuran (THF) under reflux conditions. The reaction is highly exothermic and requires careful control to prevent runaway reactions.
Applications[edit]
Diethylzinc is used in organic synthesis, primarily as a reagent for the introduction of the ethyl group into organic molecules. It acts as a strong nucleophile and can be used in the formation of carbon-zinc bonds. Diethylzinc is also employed in the synthesis of other organozinc compounds and as a reducing agent in certain types of reactions.
In addition to its use in chemical synthesis, diethylzinc has been explored as a potential reagent for the deposition of zinc-containing films in materials science applications, particularly in the area of thin-film semiconductors and nanotechnology.
Safety[edit]
Due to its pyrophoric nature and the potential for violent reaction with water, handling diethylzinc requires strict safety precautions. It should be stored under an inert atmosphere in tightly sealed containers. Protective equipment, including gloves and eye protection, is essential when working with this compound. In case of a spill or accidental exposure, appropriate emergency procedures should be followed to mitigate the risk of fire or chemical burns.
See also[edit]
References[edit]
-
Structure of Diethylzinc
-
3D ball model of Diethylzinc
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian