Diphenylmethane
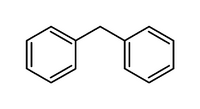
Diphenylmethane is an organic compound with the formula (C6H5)2CH2. The molecule consists of two phenyl groups (C6H5) bonded to a central methylene group (-CH2-). It is a colorless solid that is used as a precursor to other organic compounds.
Structure and synthesis[edit]
Diphenylmethane is a simple aromatic compound that consists of two phenyl groups attached to a central methylene group. The molecule is planar, with the two phenyl rings oriented at an angle of 120 degrees to each other. The carbon-carbon bond length between the methylene group and the phenyl rings is approximately 1.5 angstroms.
Diphenylmethane can be synthesized by the reaction of benzyl chloride with phenylmagnesium bromide, a Grignard reagent. The reaction proceeds via a nucleophilic addition mechanism, with the Grignard reagent attacking the electrophilic carbon of the benzyl chloride.
Uses[edit]
Diphenylmethane is primarily used as a precursor to other organic compounds. It is a starting material in the synthesis of diphenylmethane diisocyanate (MDI), a key component in the production of polyurethane foams. Diphenylmethane is also used in the synthesis of diphenylmethane diamine, a curing agent for epoxy resins.
Safety[edit]
Diphenylmethane is not considered highly toxic, but it can cause irritation to the skin and eyes. It is also harmful if swallowed or inhaled. Proper safety precautions should be taken when handling this compound, including the use of personal protective equipment and adequate ventilation.
See also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
