EIDD-1723
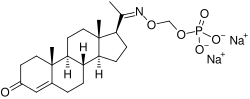
EIDD-1723 is an antiviral drug candidate that is being developed for the treatment of COVID-19. It is a prodrug of the active compound EIDD-2801, which is metabolized in the body to N4-hydroxycytidine, a nucleoside analog that inhibits viral RNA polymerase.
History[edit]
EIDD-1723 was first synthesized by researchers at Emory University's Drug Innovation Ventures (DRIVE). The compound was later licensed to Ridgeback Biotherapeutics, a Miami-based biotechnology company, for further development and clinical testing.
Mechanism of Action[edit]
EIDD-1723 works by introducing copying errors into the viral genome during replication. The active metabolite, N4-hydroxycytidine, is incorporated into the viral RNA by the viral RNA polymerase. This causes a high rate of mutation in the viral genome, leading to a phenomenon known as "error catastrophe" or "lethal mutagenesis".
Clinical Trials[edit]
In 2020, EIDD-1723 entered Phase 1 clinical trials for safety and pharmacokinetics in healthy volunteers. The drug is also being tested in animal models for efficacy against a range of viruses, including SARS-CoV-2, the virus that causes COVID-19.
Potential Applications[edit]
In addition to its potential use in treating COVID-19, EIDD-1723 may also have applications in the treatment of other viral diseases. Preclinical studies have shown that the drug has broad-spectrum antiviral activity against a range of RNA viruses, including influenza, Ebola, and coronaviruses.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian