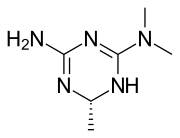
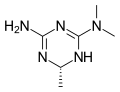
Imeglimin
An oral antidiabetic medication
| Imeglimin | |
|---|---|
|
|
Imeglimin is an oral antidiabetic medication used in the management of type 2 diabetes mellitus. It belongs to a novel class of oral hypoglycemic agents known as the glimins. Imeglimin is unique in its mechanism of action, targeting mitochondrial bioenergetics to improve insulin sensitivity and reduce hepatic glucose production.
Mechanism of Action[edit]
Imeglimin acts by modulating mitochondrial function, which is crucial in the pathophysiology of type 2 diabetes. It enhances insulin sensitivity in muscle tissue, reduces hepatic glucose output, and improves pancreatic beta-cell function. This multifaceted approach helps in maintaining blood glucose levels within the normal range.
Pharmacokinetics[edit]
Imeglimin is administered orally and is absorbed in the gastrointestinal tract. It undergoes minimal metabolism and is primarily excreted unchanged in the urine. The pharmacokinetic profile of imeglimin supports its use as a twice-daily medication, providing sustained glucose control throughout the day.
Clinical Use[edit]
Imeglimin is indicated for the treatment of type 2 diabetes, particularly in patients who have not achieved adequate glycemic control with other oral antidiabetic agents. It can be used as monotherapy or in combination with other medications such as metformin, sulfonylureas, or DPP-4 inhibitors.
Side Effects[edit]
Common side effects of imeglimin include gastrointestinal disturbances such as nausea and diarrhea. These side effects are generally mild and tend to resolve with continued use. Imeglimin is well-tolerated, with a safety profile similar to other oral antidiabetic agents.
Development and Approval[edit]
Imeglimin was developed by Poxel, a pharmaceutical company specializing in metabolic disorders. It has undergone extensive clinical trials to evaluate its efficacy and safety in patients with type 2 diabetes. Imeglimin has been approved for use in several countries and is undergoing further evaluation for approval in additional markets.
Related pages[edit]
-
Imeglimin
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian