Incyclinide
An overview of the antibiotic incyclinide
| Incyclinide | |
|---|---|
|
|
Incyclinide is a synthetic antibiotic belonging to the class of tetracycline antibiotics. It is primarily used for its antimicrobial properties and has been studied for various therapeutic applications.
Chemical Structure[edit]
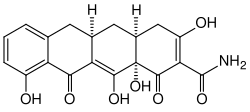
Incyclinide is characterized by its complex chemical structure, which is typical of tetracycline derivatives. The structure includes multiple ring systems and functional groups that contribute to its pharmacological activity.
Mechanism of Action[edit]
Incyclinide functions by inhibiting protein synthesis in bacteria. It binds to the 30S ribosomal subunit, preventing the attachment of aminoacyl-tRNA to the mRNA-ribosome complex. This action effectively halts bacterial growth, making it a bacteriostatic agent.
Therapeutic Uses[edit]
Incyclinide has been explored for use in treating various bacterial infections. Its efficacy against gram-positive and gram-negative bacteria makes it a versatile option in antibiotic therapy. Additionally, research is ongoing to evaluate its potential in treating inflammatory diseases and certain cancers.
Pharmacokinetics[edit]
The absorption, distribution, metabolism, and excretion of incyclinide are similar to other tetracyclines. It is well-absorbed orally and widely distributed throughout the body. The drug is metabolized in the liver and excreted primarily via the kidneys.
Side Effects[edit]
Common side effects of incyclinide include gastrointestinal disturbances, such as nausea and diarrhea. Like other tetracyclines, it can cause photosensitivity and discoloration of teeth if used in children or during pregnancy.
Resistance[edit]
Bacterial resistance to incyclinide can occur through various mechanisms, including efflux pumps and ribosomal protection proteins. The development of resistance is a significant concern in the clinical use of tetracyclines.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian