Isopentane
Isopentane[edit]
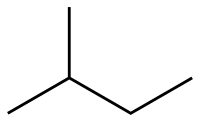
Isopentane, also known as 2-methylbutane, is a branched alkane with the chemical formula C5H12. It is one of the three structural isomers of pentane, the others being n-pentane and neopentane. Isopentane is a colorless, flammable liquid at room temperature and is commonly used in the petrochemical industry.
Structure and Properties[edit]
Isopentane is characterized by its branched structure, where a methyl group (CH3) is attached to the second carbon of a butane chain. This branching results in a lower boiling point compared to its straight-chain isomer, n-pentane. Isopentane has a boiling point of approximately 27.8 °C (82.0 °F) and a melting point of -159.9 °C (-255.8 °F).
The molecular geometry of isopentane is tetrahedral around each carbon atom, typical of alkanes. The presence of branching in isopentane reduces the van der Waals forces between molecules, leading to its lower boiling point.
Production[edit]
Isopentane is primarily obtained from the fractional distillation of crude oil and natural gas. It is often found in the light naphtha fraction and can be separated through distillation processes. Additionally, isopentane can be synthesized through the isomerization of n-pentane using catalysts such as platinum or zeolites.
Uses[edit]
Isopentane is widely used as a blowing agent in the production of polystyrene and other foam products. Its low boiling point makes it ideal for this application, as it vaporizes quickly, creating a cellular structure in the foam.
In the petroleum industry, isopentane is used as a solvent and as a component in gasoline blends to increase the octane rating. It is also used in geothermal power plants as a working fluid in binary cycle power plants due to its favorable thermodynamic properties.
Safety and Environmental Impact[edit]
Isopentane is highly flammable and poses a risk of fire and explosion. It should be handled with care, and appropriate safety measures should be in place when working with this chemical.
In terms of environmental impact, isopentane is a volatile organic compound (VOC) and can contribute to the formation of ground-level ozone and smog. However, it is not considered a significant greenhouse gas.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian