Menthol
Organic compound used as flavoring and in medicinal applications
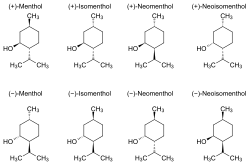
Menthol is an organic compound made synthetically or obtained from the oils of corn mint, peppermint, or other mints. It is a waxy, crystalline substance, clear or white in color, which is solid at room temperature and melts slightly above. The main form of menthol occurring in nature is (−)-menthol, which is assigned the (1R,2S,5R) configuration.
Chemical Structure and Properties[edit]
Menthol is a terpene alcohol with the chemical formula C10H20O. It has three asymmetric carbon atoms in the cyclohexane ring, which allows for the existence of eight stereoisomers. The most common isomer in nature is (−)-menthol, which has a specific configuration that contributes to its characteristic cooling sensation.
Menthol is known for its ability to trigger the cold-sensitive TRPM8 receptors in the skin, which is why it is often used in products designed to provide a cooling sensation.
Sources and Production[edit]
Menthol can be extracted from mint oils, such as peppermint oil, or it can be synthesized. The synthetic production of menthol involves several chemical processes, including the Haarmann-Reimer process, which is a well-known method for industrial synthesis.
Natural Sources[edit]
Menthol is primarily obtained from the essential oils of mint plants, particularly peppermint (Mentha piperita) and corn mint (Mentha arvensis). These plants are cultivated in various regions around the world, with India being one of the largest producers of natural menthol.
Synthetic Production[edit]
Synthetic menthol is produced through several methods, including the hydrogenation of thymol and the Haarmann-Reimer process. These methods allow for the large-scale production of menthol to meet global demand.
Applications[edit]
Menthol is widely used in various applications due to its cooling properties and pleasant aroma. It is a common ingredient in oral hygiene products, cough drops, topical analgesics, and flavoring agents in food and beverages.
Medicinal Uses[edit]
In medicine, menthol is used as a topical analgesic to relieve minor aches and pains. It is also used in cough suppressants and decongestants due to its ability to provide a cooling sensation and relieve throat irritation.
Flavoring and Fragrance[edit]
Menthol is used as a flavoring agent in chewing gum, candy, and toothpaste. Its refreshing aroma makes it a popular choice in perfumes and cosmetics.
Biosynthesis[edit]
The biosynthesis of menthol in plants involves several enzymatic steps starting from geranyl diphosphate, a common precursor in the biosynthesis of monoterpenes.
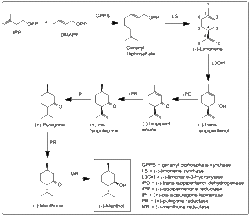
Physical Properties[edit]
Menthol is a crystalline substance that is solid at room temperature. It has a melting point of approximately 36 °C (97 °F) and a boiling point of 212 °C (414 °F). It is slightly soluble in water but highly soluble in organic solvents such as alcohol and ether.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian