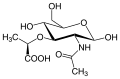
N-Acetylmuramic acid
N-Acetylmuramic acid (also known as MurNAc or NAM) is a derivative of glucosamine and a key component in the peptidoglycan layer of bacterial cell walls. It is a unique monosaccharide that is not found in eukaryotic cells, making it a target for antibiotic drugs.
Structure and Synthesis[edit]
N-Acetylmuramic acid is a monosaccharide composed of a glucose molecule with an acetyl group and a lactic acid molecule attached. The synthesis of N-Acetylmuramic acid begins with the conversion of fructose-6-phosphate to glucosamine-6-phosphate by the enzyme glutamine-fructose-6-phosphate aminotransferase. This is followed by the addition of an acetyl group to form N-acetylglucosamine-6-phosphate, and finally the addition of a lactic acid molecule to form N-Acetylmuramic acid.
Role in Bacterial Cell Walls[edit]
N-Acetylmuramic acid is a crucial component of the peptidoglycan layer in bacterial cell walls. It forms a disaccharide with N-acetylglucosamine (NAG), and these disaccharides are linked together by beta-1,4-glycosidic bonds to form a polysaccharide chain. These chains are cross-linked by short peptide chains to form the rigid peptidoglycan layer that provides structural support to the bacterial cell.
Antibiotic Target[edit]
Because N-Acetylmuramic acid is unique to bacterial cells and crucial for their survival, it is a target for antibiotic drugs. Penicillin and other beta-lactam antibiotics work by inhibiting the enzymes that cross-link the peptidoglycan chains, preventing the formation of a functional cell wall and leading to the death of the bacterial cell.
See Also[edit]
N-Acetylmuramic acid gallery[edit]
-
N-Acetylmuramic acid
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian