N-Nitroso-N-methylurea
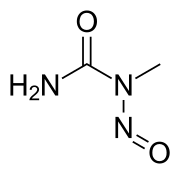
N-Nitroso-N-methylurea (NMU) is a highly potent carcinogen widely used in biochemical research. It is a derivative of urea, with a nitroso group and a methyl group replacing two hydrogen atoms.
Chemical Properties[edit]
NMU is a yellow, crystalline solid with a melting point of 89-92°C. It is soluble in water and most organic solvents. It is highly reactive, decomposing in the presence of light, heat, or strong acids to release nitric oxide and methylamine.
Biological Effects[edit]
NMU is a potent carcinogen, capable of inducing tumors in a variety of tissues in many species. It acts by alkylating DNA, leading to the formation of O6-methylguanine lesions, which can cause mutations if not repaired by the DNA repair machinery of the cell.
Use in Research[edit]
NMU is widely used in biochemical research to induce tumors in experimental animals, particularly rats and mice. It is often used in studies of carcinogenesis, mutagenesis, and DNA repair.
Safety[edit]
Due to its high carcinogenic potency, NMU must be handled with extreme care. It is classified as a Group 1 carcinogen by the International Agency for Research on Cancer (IARC), meaning it is known to cause cancer in humans.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
