Pyrrole
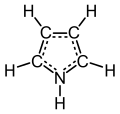
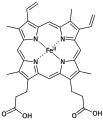
Pyrrole is a heterocyclic aromatic organic compound, a five-membered ring with the formula C4H4NH. It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., N-methylpyrrole, C4H4NCH3. Porphobilinogen, a trisubstituted pyrrole, is the biosynthetic precursor to many natural products such as heme.
Structure and bonding[edit]
Pyrroles are aromatic. Like furan and thiophene, the pyrrole ring is aromatic because it contains 4 π-electrons in the two double bonds plus one lone pair of electrons on the nitrogen. The lone pair on the nitrogen is not part of the aromatic π-system. Pyrroles are weakly basic, with a conjugate acid pKa of −3.8. The most basic site is C-2. Pyrroles are also weakly acidic at the N-H position, with a pKa of 17.5.
Preparation[edit]
Pyrrole can be prepared by treatment of furan with ammonia in the presence of acid catalysts, such as AlCl3. Pyrrole can also be formed by dehydrogenation of pyrrolidine.
Reactions[edit]
Pyrrole undergoes reactions that are characteristic of other aromatic compounds, notably electrophilic aromatic substitution. It is particularly susceptible to such reactions because of the electron-donating effect of the nitrogen. Hence, electrophiles attack pyrrole at the 2 position, then rearrangement of the intermediate σ-complex leads to substitution at the 3 position.
Applications[edit]
Pyrrole is a constituent of tobacco smoke and not as an ingredient. Pyrrole is used as a precursor to the drug tolmetin. Polypyrrole is of some commercial value.
See also[edit]
References[edit]
Pyrrole[edit]
-
Pyrrole 2D full
-
Pyrrole 2D numbered
-
Pyrrole CRC MW 3D balls A
-
Pyrrole CRC MW 3D vdW
-
Heme B
-
Pyrrolsynthese 1
-
Hantzsch Pyrrole Synthesis Scheme
-
Knorr Pyrrole Synthesis Scheme
-
Paal-Knorr Pyrrole Synthesis
-
Van Leusen Mechanism
-
Barton-Zard reaction
-
Piloty-Robinson reaction
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian