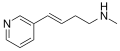
Rivanicline
Rivanicline (also known as ABT-894) is a drug that was developed by Abbott Laboratories for the treatment of tobacco dependence and neuropathic pain. It acts as a nicotinic acetylcholine receptor partial agonist, similar to the smoking cessation drug varenicline.
History[edit]
Rivanicline was developed by Abbott Laboratories, a global healthcare company, in the early 2000s. The drug was initially designed to treat tobacco dependence and neuropathic pain, two conditions that affect millions of people worldwide.
Mechanism of Action[edit]
Rivanicline works by acting as a partial agonist at the nicotinic acetylcholine receptor. This means that it partially activates these receptors, which are involved in the addictive effects of nicotine. By doing so, it can help to reduce cravings for nicotine and ease withdrawal symptoms.
Comparison with Varenicline[edit]
Rivanicline is similar in action to varenicline, another drug used to treat tobacco dependence. Both drugs act as partial agonists at the nicotinic acetylcholine receptor. However, there may be differences in their efficacy and side effect profiles, which could make one drug more suitable than the other for certain patients.
Clinical Trials[edit]
Rivanicline has undergone clinical trials to assess its safety and efficacy in treating tobacco dependence and neuropathic pain. However, as of 2021, it has not been approved for use by any major regulatory bodies.
Side Effects[edit]
As with any drug, rivanicline may cause side effects. These can include nausea, headache, and sleep disturbances. It is important for patients to discuss these potential side effects with their healthcare provider before starting treatment.
Future Research[edit]
Future research on rivanicline may focus on further elucidating its mechanism of action, optimizing its dosing regimen, and comparing its efficacy and safety to other treatments for tobacco dependence and neuropathic pain.
-
Rivanicline
-
Rivanicline
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian