Sulfur dioxide
Sulfur dioxide (chemical formula: SO2) is a chemical compound that is a toxic gas with a pungent, irritating smell. It is released naturally by volcanic activity and is produced as a by-product of the burning of fossil fuels contaminated with sulfur compounds.
Chemical properties[edit]
Sulfur dioxide is a bent molecule with C2v symmetry point group. A significant amount of the sulfur dioxide produced in the atmosphere is the result of the reaction between sulfur and oxygen.
Uses[edit]
Sulfur dioxide is an important compound in many industries. It is used in the production of sulfuric acid, one of the most important chemicals manufactured by the chemical industry. It is also used as a preservative in some foods, particularly dried fruits, and as a reducing agent in brewing and winemaking.
Health effects[edit]
Exposure to sulfur dioxide can have various health effects. It can cause a burning sensation in the nose and throat, difficulty breathing, and severe airway obstructions. Chronic exposure to sulfur dioxide can cause respiratory problems and alterations in the lungs' defenses.
Environmental impact[edit]
Sulfur dioxide is one of the major air pollutants and a significant contributor to acid rain. When it reacts with water, it forms sulfurous acid, which is a component of acid rain.
See also[edit]
-
Sulfur dioxide 3D vdW
-
Sulfur dioxide resonance 2D
-
Io Aurorae color
-
Burning sulfur in oxygen atmosphere
-
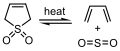
Cheletropic reaction of butadiene with SO2
-
Preparation of m-trifluoromethylbenzenesulfonyl chloride
-
Leilani Estates Hawaii Volcanic Eruption
-
Volcanic injection
-
Acid rain woods
-
Estimates of past and future SO2 global anthropogenic emissions
-
Physical Drivers of climate change
-
SPICE SRM overview
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian