Technetium (99mTc) medronic acid
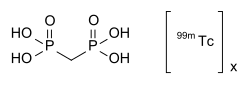
Radiopharmaceutical used in bone imaging
| Technetium (99mTc) medronic acid
| |
|---|---|
| Routes | Intravenous
|
Technetium (99mTc) medronic acid is a radiopharmaceutical used primarily in nuclear medicine for bone imaging. It is a compound that combines technetium-99m (99mTc), a widely used radioisotope, with medronic acid, a phosphonate compound that targets bone tissue.
Mechanism of Action[edit]
Technetium (99mTc) medronic acid works by binding to the hydroxyapatite crystals in bone. The technetium-99m emits gamma radiation, which can be detected by a gamma camera. This allows for the visualization of bone structure and the identification of abnormalities such as bone metastases, fractures, and osteomyelitis.
Clinical Use[edit]
Technetium (99mTc) medronic acid is used in bone scintigraphy, a diagnostic imaging technique. It is particularly useful for detecting bone metastases in patients with cancer, evaluating unexplained bone pain, and diagnosing osteomyelitis.
Administration[edit]
The compound is administered intravenously. After injection, it typically takes about 2-4 hours for optimal imaging, as the radiopharmaceutical accumulates in the bone tissue.
Safety and Side Effects[edit]
Technetium (99mTc) medronic acid is generally well-tolerated. However, potential side effects include allergic reactions, injection site reactions, and, rarely, radiation exposure risks. It is contraindicated in patients with known hypersensitivity to any component of the formulation.
Related Pages[edit]
Categories[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian