Telbivudine
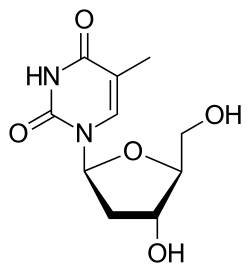
Telbivudine is an antiviral drug used in the treatment of hepatitis B. It is a synthetic thymidine nucleoside analogue with potent and selective activity against the hepatitis B virus (HBV). Telbivudine is marketed under the brand name Tyzeka in the United States and Sebivo in other countries.
Mechanism of Action[edit]
Telbivudine works by inhibiting the DNA polymerase of the hepatitis B virus, which is essential for viral replication. By incorporating itself into the viral DNA, telbivudine causes premature chain termination, thereby preventing the virus from multiplying and spreading.
Pharmacokinetics[edit]
Telbivudine is administered orally and is well absorbed from the gastrointestinal tract. It reaches peak plasma concentrations within 1 to 4 hours after ingestion. The drug is primarily excreted unchanged in the urine, with a half-life of approximately 15 hours.
Clinical Use[edit]
Telbivudine is indicated for the treatment of chronic hepatitis B in patients with evidence of active viral replication and either evidence of persistent elevations in serum alanine aminotransferase (ALT) or histologically active disease. It is particularly effective in patients who are nucleoside analogue-naive.
Side Effects[edit]
Common side effects of telbivudine include:
Resistance[edit]
Resistance to telbivudine can develop, particularly with long-term use. The most common mutation associated with resistance is the M204I mutation in the HBV polymerase gene. Cross-resistance with other nucleoside analogues, such as lamivudine and entecavir, can also occur.
Contraindications[edit]
Telbivudine is contraindicated in patients with a known hypersensitivity to the drug or any of its components. Caution is advised in patients with renal impairment, as dose adjustments may be necessary.
Pregnancy and Lactation[edit]
Telbivudine is classified as a pregnancy category B drug. It should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is not known whether telbivudine is excreted in human milk; therefore, caution should be exercised when administered to nursing mothers.
See Also[edit]
References[edit]
External Links[edit]
| Telbivudine | |
|---|---|
|
| |
| Trade names | Tyzeka, Sebivo
|
| Chemical nomenclature | |
| IUPAC name | 1-[(2S,4R,5S)-4-Hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione
|
| Routes | Oral |
| Pregnancy category | B
|
| Half-life | 15 hours |
| Excretion | Renal |
| Legal status | Rx-only |
| Identifiers | |
| CAS Number | 3424-98-4 |
| PubChem | 457898 |
| DrugBank | DB01265 |
| ChemSpider | 403168 |
| UNII | 3O2K1G66ZY |
| KEGG | D06067 |
| ChEMBL | 1201187 |
| Chemical data
| |
| Chemical formula | C10H14N2O5
|
| SMILES | CC1=CN(C(=O)NC1=O)C2CC(C(O2)CO)O
|
| InChI | 1S/C10H14N2O5/c1-5-2-12(10(16)11-9(5)15)8-3-6(14)7(4-13)17-8/h2,6-8,13-14H,3-4H2,1H3,(H,11,15,16)/t6-,7+,8+/m0/s1
|
| InChIKey | HNVIMBABAJGQLR-RRKCRQDMSA-N
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian