Trichloronate
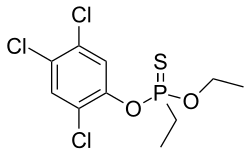
0Trichloronate is an organophosphate compound that is used as an insecticide. It is a colorless liquid with a mild odor and is highly toxic to both humans and animals.
Chemical Properties[edit]
Trichloronate is a chlorinated derivative of phosphoric acid. It has the chemical formula C3H6Cl3O4P and a molecular weight of 257.42 g/mol. It is soluble in water and most organic solvents.
Uses[edit]
Trichloronate is primarily used as an insecticide in agriculture. It is effective against a wide range of insects, including aphids, mites, and whiteflies. It is also used in the control of mosquitoes and other flying insects in public health applications.
Toxicity[edit]
Trichloronate is highly toxic to humans and animals. Exposure can occur through inhalation, ingestion, or skin contact. Symptoms of exposure include nausea, vomiting, diarrhea, abdominal cramps, sweating, blurred vision, and difficulty breathing. In severe cases, exposure can lead to seizures, loss of consciousness, and even death.
Environmental Impact[edit]
Trichloronate is highly toxic to aquatic life and can cause long-term damage to the environment if released in large quantities. It is also persistent in the environment and can accumulate in the food chain.
Regulation[edit]
In many countries, the use of trichloronate is regulated due to its high toxicity and environmental impact. Users are required to follow strict safety guidelines to minimize exposure and environmental contamination.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian

