Triphenylmethanol
Triphenylmethanol[edit]
Triphenylmethanol is an organic compound with the formula (C_H_)_COH. It is a white crystalline solid that is soluble in organic solvents but not in water. It is a member of the class of compounds known as alcohols, specifically a tertiary alcohol, due to the presence of the hydroxyl group (-OH) attached to a carbon atom that is connected to three phenyl groups.
Structure and Properties[edit]
Triphenylmethanol has a central carbon atom bonded to three phenyl groups and one hydroxyl group. This structure gives it a high degree of symmetry and stability. The compound is known for its relatively high melting point and its ability to form stable crystals. The presence of the bulky phenyl groups around the central carbon atom makes it less reactive than other alcohols.
Synthesis[edit]
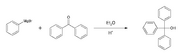
Triphenylmethanol can be synthesized through the Grignard reaction, which involves the reaction of phenylmagnesium bromide with benzophenone. The reaction proceeds as follows:
In this reaction, phenylmagnesium bromide acts as a nucleophile, attacking the carbonyl carbon of benzophenone, leading to the formation of triphenylmethanol after hydrolysis.
Applications[edit]
Triphenylmethanol is primarily used in organic chemistry as a reagent and as an intermediate in the synthesis of other compounds. It is also used in the study of reaction mechanisms due to its stability and well-defined structure.
Related Compounds[edit]
Triphenylmethanol is related to other triphenylmethyl compounds, such as triphenylmethyl chloride and triphenylmethyl radical. These compounds share the triphenylmethyl group but differ in their functional groups and reactivity.
Related Pages[edit]
Triphenylmethanol[edit]
-
Synthesis of triphenylmethanol
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian